GLOBAL Advocacy – Elongating Life & Improving Health

What we do
Policy and Advocacy
Featured Action

Urge Congress to pass the DeOndra Dixon INCLUDE Project Act
This bill will protect and help increase funding for life-saving, transformative Down syndrome research and medical care at the National Institutes of Health
Our Impact
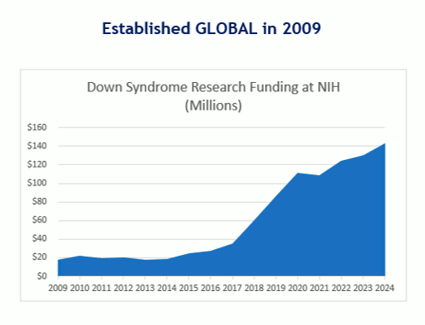 GLOBAL was established in 2009 specifically to address the life-threatening lack of research and medical care for people with Down syndrome at our National Institutes of Health (NIH). For nearly two decades, Down syndrome was one of the least funded genetic conditions at NIH.
GLOBAL was established in 2009 specifically to address the life-threatening lack of research and medical care for people with Down syndrome at our National Institutes of Health (NIH). For nearly two decades, Down syndrome was one of the least funded genetic conditions at NIH.
With support from our families, scientists, medical professionals, and Congressional and NIH Champions, GLOBAL has successfully lobbied and advocated for the NIH to create a “trans-NIH” Down syndrome funding project called “INCLUDE” which draws funding from 18 of NIH’s 27 different institutes and centers.
In just six years our advocacy has added $338 million to life-changing Down syndrome research at the NIH through the INCLUDE Project. With your help, we’ll get to our goal of $250 million a year which is on par with autism.
Urge Congress to pass the DeOndra Dixon INCLUDE Project Act
This bill will protect and help increase funding for life-saving, transformative Down syndrome research and medical care at the National Institutes of Health
All the impactful research studies that have been funded by NIH INCLUDE Project are organized for easy public access by the NIH INCLUDE Data Coordinating Center which is co-managed by GLOBAL’s Affiliate, the Crnic Institute for Down syndrome.
